COVID-19 during flu season; doctors urging Americans to get updated vaccine
In updated guidance, the Centers for Disease Control and Prevention says anyone 6 months and older can now get vaccinated for COVID-19. This month, the FDA approved an updated booster to target new variants.
In updated guidance, the Centers for Disease Control and Prevention says anyone 6 months and older can now get vaccinated for COVID-19. This month, the FDA approved an updated booster to target new variants.
US approves updated COVID vaccines to rev up protection this fall
The U.S. approved updated COVID-19 vaccines Monday, hoping to rev up protection against the latest coronavirus strains and blunt any surge this fall and winter.
The U.S. approved updated COVID-19 vaccines Monday, hoping to rev up protection against the latest coronavirus strains and blunt any surge this fall and winter.
Covid cases increasing in every state, Texas listed among the highest in a new study
A new report showing the covid cases in every state, ranked Texas as one of the highest in the country.
A new report showing the covid cases in every state, ranked Texas as one of the highest in the country.
Harris County Public Heath to offer COVID booster shots to children 6 months, older
Harris County Public Health will administer updated COVID-19 booster shots to children 6 months and older on Friday.
Harris County Public Health will administer updated COVID-19 booster shots to children 6 months and older on Friday.
US health officials renew push for COVID-19 boosters, amid new data on efficacy
Only about 13% of U.S. adults have gotten the updated booster.
Only about 13% of U.S. adults have gotten the updated booster.
Pfizer says COVID-19 vaccine will cost $110-$130 per dose
Pfizer said the commercial pricing for adult COVID-19 vaccine doses could start in early 2023, depending on when the U.S. government phases out its program of buying the shots.
Pfizer said the commercial pricing for adult COVID-19 vaccine doses could start in early 2023, depending on when the U.S. government phases out its program of buying the shots.
FDA OKs Novavax COVID-19 booster dose
The Food and Drug Administration has authorized a booster dose of the COVID-19 vaccine made by Novavax.
The Food and Drug Administration has authorized a booster dose of the COVID-19 vaccine made by Novavax.
White House: Get new COVID booster by Halloween
The White House is warning of a "challenging" virus season ahead and urging eligible Americans to get the updated COVID-19 boosters by Halloween to have maximum protection against the coronavirus by Thanksgiving and the holidays
The White House is warning of a "challenging" virus season ahead and urging eligible Americans to get the updated COVID-19 boosters by Halloween to have maximum protection against the coronavirus by Thanksgiving and the holidays
Should I get the new COVID booster and flu shot at the same time? Your questions, answered
With flu season approaching in the U.S., as well as the availability of the new omicron-designed COVID-19 booster for everyone 12 and up, here are some answers to common questions about the shots.
With flu season approaching in the U.S., as well as the availability of the new omicron-designed COVID-19 booster for everyone 12 and up, here are some answers to common questions about the shots.
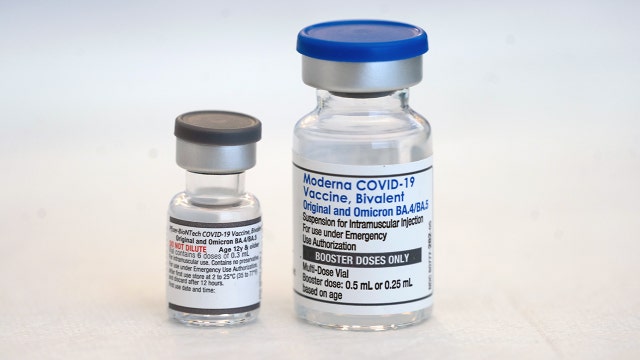
COVID-19 bivalent shots targeting other variants making their way to Houston
Health officials recently gave emergency authorization for the COVID-19 ‘combo shots’ to help combat variants of the coronavirus and eligible Houston residents can soon get theirs.
Health officials recently gave emergency authorization for the COVID-19 ‘combo shots’ to help combat variants of the coronavirus and eligible Houston residents can soon get theirs.
CDC panel recommends tweaked COVID-19 booster
The tweaked COVID booster, which was approved in a fast-tracked strategy, is expected to be available next week after Labor Day.
The tweaked COVID booster, which was approved in a fast-tracked strategy, is expected to be available next week after Labor Day.
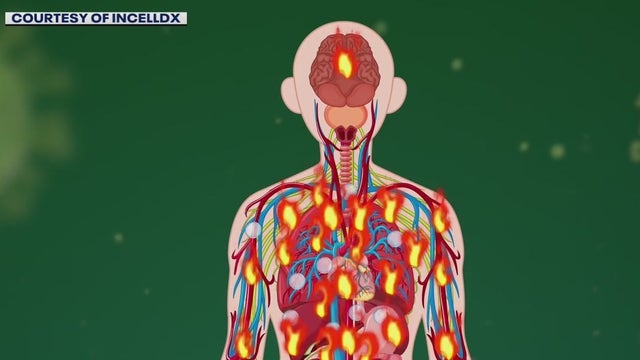
New test and treatments for patients with lingering COVID-19 symptoms
Known as long-haulers, many patients aren't sure if COVID-19 is truly what they are suffering from, but now a simple blood test may provide much-needed answers, along with a treatment plan.
Known as long-haulers, many patients aren't sure if COVID-19 is truly what they are suffering from, but now a simple blood test may provide much-needed answers, along with a treatment plan.
Harris County Public Health offering COVID-19 Novavax vaccine to kids ages 12 to 17
Harris County Public Health is now offering the COVID-19 Novavax vaccine to kids ages 12 to 17. Novavax is a two-dose primary series given three weeks apart.
Harris County Public Health is now offering the COVID-19 Novavax vaccine to kids ages 12 to 17. Novavax is a two-dose primary series given three weeks apart.
CDC drops quarantine, screening recommendations for COVID-19
The nation's top public health agency on Thursday relaxed its COVID-19 guidelines, dropping the recommendation that Americans quarantine themselves if they come into close contact with an infected person.
The nation's top public health agency on Thursday relaxed its COVID-19 guidelines, dropping the recommendation that Americans quarantine themselves if they come into close contact with an infected person.
Biden's latest COVID-19 'rebound' case has researchers evaluating link to anti-viral drug
Health experts say during clinical trials, rebound cases happened in only 1 to 2% of people.
Health experts say during clinical trials, rebound cases happened in only 1 to 2% of people.
Texas Southern University offers free vaccinations for students
In partnership with the Houston Health Department, TSU is offering three vaccines including meningitis, flu, and COVID-19 for free.
In partnership with the Houston Health Department, TSU is offering three vaccines including meningitis, flu, and COVID-19 for free.
NIH: COVID-19 booster-induced antibody response to omicron significantly decreases over time
Researchers found that while COVID-19 booster vaccinations create a strong antibody response against omicron initially, within three months post-boost, those levels decrease.
Researchers found that while COVID-19 booster vaccinations create a strong antibody response against omicron initially, within three months post-boost, those levels decrease.
EU urges second COVID-19 boosters for people ages 60 to 79
The European Union said Monday it's “critical” that authorities in the 27-nation bloc consider giving second coronavirus booster shots as a new wave of the pandemic sweeps over the continent.
The European Union said Monday it's “critical” that authorities in the 27-nation bloc consider giving second coronavirus booster shots as a new wave of the pandemic sweeps over the continent.
COVID-19 omicron subvariants BA.4 and BA.5 could be ‘worst’ so far, doctors say
Subvariants BA.4 and BA.5 are more transmissible, and early evidence shows both are capable of evading immune responses from previous infection as well as current COVID-19 vaccines.
Subvariants BA.4 and BA.5 are more transmissible, and early evidence shows both are capable of evading immune responses from previous infection as well as current COVID-19 vaccines.
New vaccine could protect against COVID-19 variants before they emerge
The vaccine works by using spike proteins from SARS-CoV-2 — the virus that causes COVID-19 — and seven other related animal viruses with the potential to cause a pandemic in humans.
The vaccine works by using spike proteins from SARS-CoV-2 — the virus that causes COVID-19 — and seven other related animal viruses with the potential to cause a pandemic in humans.